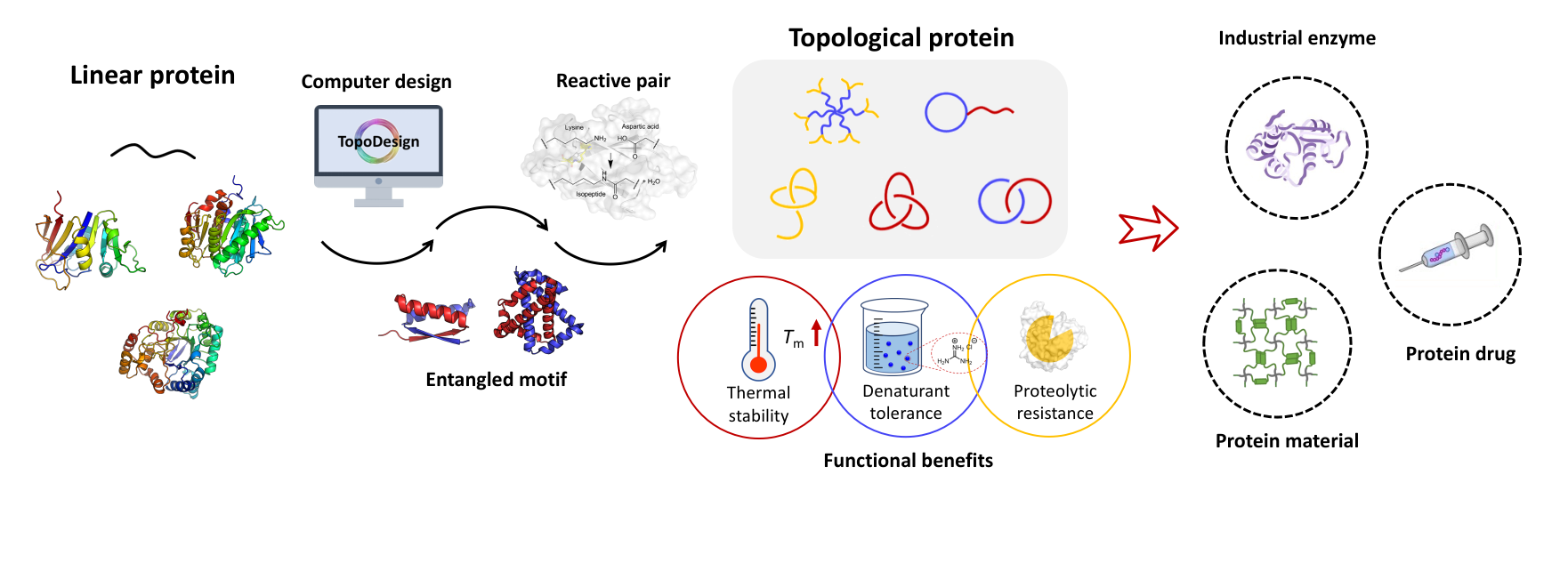
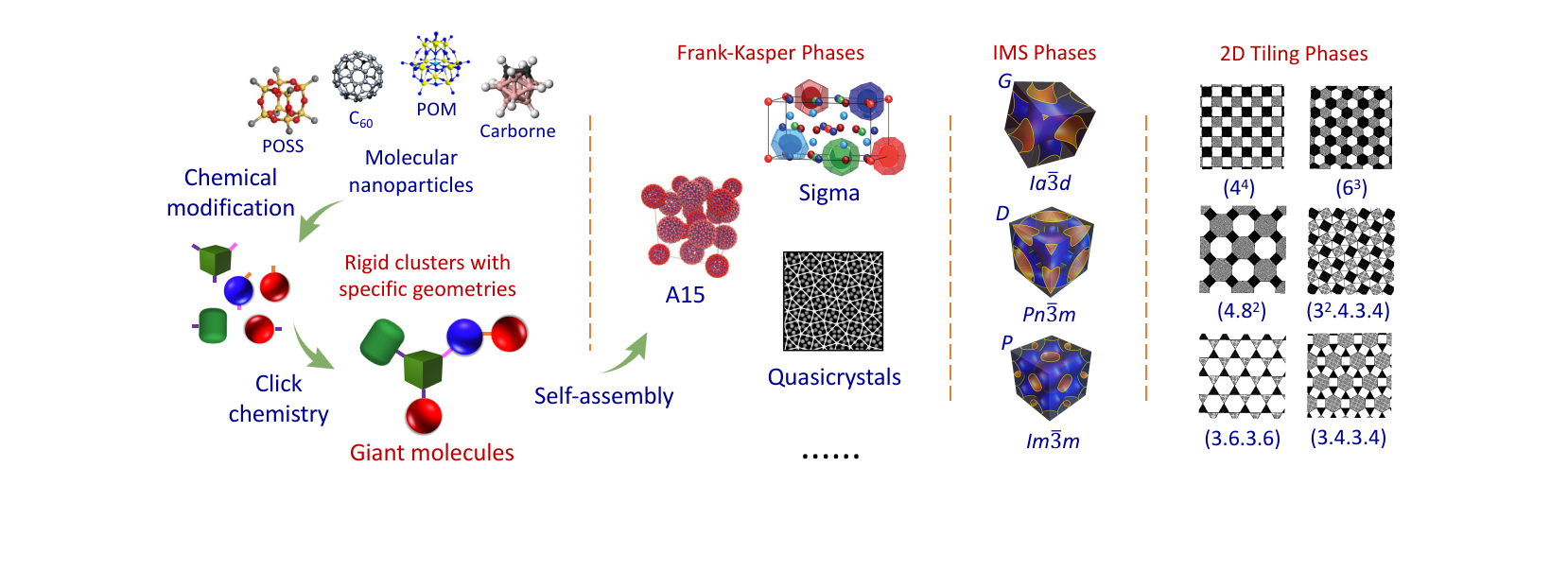
箴言
----------------------------------------------
-----------------------------------------------
在科学上没有平坦的大道,只有那些不畏艰险沿着陡峭山路攀登的人,才有希望达到光辉的顶点。
----马克思
-----------------------------------------------
合作研究
------------------------------------------
请有兴趣的研究组联系我们。欢迎任何形式的合作,尤其是在自组装、水凝胶以及生物医药等方向的合作。
------------------------------------------
请有兴趣的研究组联系我们。欢迎任何形式的合作,尤其是在自组装、水凝胶以及生物医药等方向的合作。
------------------------------------------
研究成果
88. Reversible Hydrogels with Tunable Mechanical Properties for Optically Controlling Cell Migration. Nano Research 2017
发布时间:2018-01-27
Wu, X.;# Huang, W.; # Wu, W.; # Xue, B.; Xiang, D.; Li, Y.; Qin, M.; Sun, F.; Wang, W.;* Zhang, W.-B.;* Cao, Y.* Reversible Hydrogels with Tunable Mechanical Properties for Optically Controlling Cell Migration. Nano Research 2018, DOI: 10.1007/s12274-017-1890-y. (#equal contribution) [Link]

Abstract
Synthetic hydrogels are widely used as biomimetic in vitro model systems to understand how cells respond to complex microenvironments. The mechanical properties of hydrogels are deterministic for many cellular behaviors, including cell migration, spreading, and differentiation. However, it remains a major challenge to engineer hydrogels that recapture the dynamic mechanical properties of native extracellular matrices. Here, we provide a new hydrogel platform with spatiotemporally tunable mechanical properties to assay and define cellular behaviors under light. The change in the mechanical properties of the hydrogel is effected by a photo-induced switch of the cross-linker fluorescent protein, Dronpa145N, between the tetrameric and monomeric states, which causes minimal changes to the chemical properties of the hydrogel. The mechanical properties can be rapidly and reversibly tuned for multiple cycles using visible light, as confirmed by rheological measurements and atomic force microscopybased nano-indentation. We further demonstrated real-time and reversible modulation of cell migration behaviors on the hydrogels through photo-induced stiffness switching, with minimal invasion to the cultured cells. Hydrogels with a programmable mechanical history and a spatially defined mechanical hierarchy might serve as an ideal model system to better understand complex cellular functions.